Amatoxin: The Deadliest of All Mushroom Poisonings
By Debbie Viess
Although amatoxin causes human and animal deaths around the world every year, with prompt care in a modern medical/veterinary facility, the chances of surviving and having a full recovery are quite good. Only around 12% of healthy adults poisoned will die or require a liver transplant. Without such care, human deaths are closer to 50%. We lack good statistics for pet poisonings, but their poisonings are often more severe and more often fatal, due to their low body weights versus volume of mushroom eaten, delay in diagnosis, and the high cost of care. But even in the case of dog poisonings, there are protocols available that can help your dog to survive. And if you can catch your dog in the act of eating an amatoxin mushroom, and begin care immediately, those odds of survival rise as well.
Which Mushrooms Contain Amatoxins?
Four genera of fungi potentially contain amatoxins and related toxins, but not all species within these genera do. There are far more harmless, or species of lesser toxicity in the genus Amanita, mostly harmless species in Lepiota, and only one toxic species currently known in Galerina. This is where the proper identification of fungal species is essential to proper diagnosis and treatment; identification to genus alone is often not enough. Although some of these known amatoxin species also contain other cyclic polypeptides like virotoxins and phallotoxins, only amatoxins cause oral poisonings.
Amanita species in section Phalloideae—big, showy and tempting—cause the vast majority of fatal mushroom poisonings around the world. They can be roughly broken into two groups: the colored “Death Caps,” like Amanita phalloides, and the white or mostly white “Destroying Angels,” like Amanita virosa and A. bisporigera. Many different species within these two groups are found all over the world.
Amanitas in section Phalloideae are covered by a membranous universal veil (UV) when young and in their “egg” state. This membrane tears as the mushroom grows and expands, leaving a loose membranous cup at the base of the stipe. The caps of these various deadly amanitas never have warts, only rarely show a thin patch of membranous UV, and normally do not have a striate cap margin, except as the mushroom ages and dries. They have free white gills and white spores. The spores are amyloid (turn blue) when immersed in Melzers solution.

Amanita phalloides, the "Death Cap," USA, Debbie Viess |

Amanita amerivirosa, "Destroying Angel," USA, John Plischke, MushroomObserver |
Small Lepiota species, up to 6 cm tall and often with pinkish tones and a sweet or fruity odor, have also caused deadly poisonings; fortunately, due to their small size they are rarely gathered as edibles. These dangerously toxic Lepiotas can be difficult to distinguish from other small, harmless species of Lepiota, which is why foragers are advised to never eat small Lepiotas.
Despite conflicting evidence published over the decades on the presence of amatoxin in a wide variety of Lepiota species, very few species have been found to contain amatoxins with modern methodology and analysis. Documented poisonings have occurred with Lepiota brunneoincarnata, L. subincarnata and L. venenata; all of these amatoxin containing species, including L. elaiophylla and A. spiculata (a large, orange-capped outlier, associated with termite mounds in the sub-tropics), form a monophyletic clade. Lepiota farinolens is also in this clade, and is likely to contain amatoxins, but was not available for testing in the most recent study. Lepiota boudieri, found outside of this amatoxin clade, was shown to contain a possible amatoxin derivative.

Lepiota brunneoincarnata, Portugal, Zaca, MushroomObserver |

Lepiota subincarnata, USA, Christian Schwarz, MushroomObserver |
 Lepiota venenata, China, http://www.facesoffungi.org |

FIGURE 1. Lepiota spiculata. (A) Basidiomes in habitat growing on a termite nest (photo by C. Angelini). (B)Representations on stamps and websites. Person in image is Claudio Angelini. Creative Commons. Copyright © 2020 Angelini, Vizzini, Justo, Bizzi, Davoli and Kaya.. |
Galerina marginata (with many former names/synonyms!) is a small brown mushroom found on wood or in wood chips, rarely in grass. There are many non-toxic Galerina species as well. They are rarely eaten deliberately, although some have mistaken them for Psilocybes, with deadly effects.
Pholiotina rugosa (formerly called Conocybe filaris) is another small, brown amatoxin containing mushroom that grows on the ground in duff. It is delicate and unlikely to be deliberately eaten. We have no records of poisonings with this species. Note: there are currently no amatoxin containing fungi known to occur in the genus Conocybe.

Galerina marginata, USA, Mike Wood, Mykoweb |

Pholiotina rugosa, USA, Fred Stevens, Mykoweb |
All of these above mushrooms can be mistaken for harmless forms by unwary hunters, and can be eaten in error by small children “grazing” in their own backyards, or by pets.
Mode of action of Amatoxin
Amatoxin is a small, complex, cyclic octapeptide with a highly stable molecular configuration, making them resistant to enzymatic action. The water-soluble amatoxin molecule is not broken down by heat, acid bath, or digestive enzymes, and is readily absorbed from the digestive tract, within 90-120 minutes post ingestion.
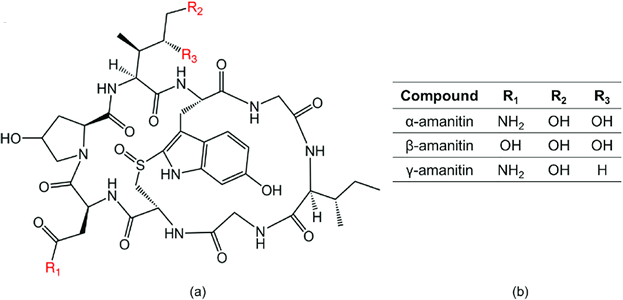
Chemical structure of amatoxin: Candace Bever (creative commons open access)
The action of amatoxin involves three cellular components: RNA polymerase II, the DNA template, and a lengthening RNA product (i.e. protein). Amatoxin binds at the site of the “swinging bridge” found in the center of the RNA polymerase II enzyme, where it physically blocks transcription and further protein synthesis (see diagram). Once bound, all protein synthesis stops, and apoptosis (cell death) occurs.
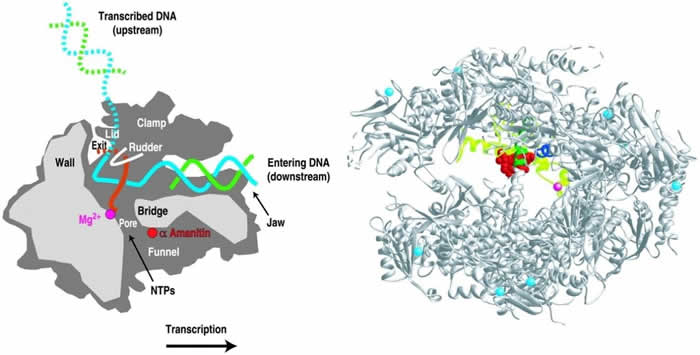
Amatoxin/RNA polymerase II: Courtesy David Bushnell
Amatoxin's greatest effects are in organs that have the fastest cell turnover: first the gut and then the liver, and sometimes, especially in cases with severe dehydration, the kidneys. The gastrointestinal (GI) tract is the first organ to be exposed. Epithelial cells lining the gut quickly absorb amatoxins. Protein synthesis is inhibited and the mucosal cells rapidly deteriorate.
Amatoxin is also readily absorbed from the digestive tract, where it is transported to the liver via the hepatic portal system/bloodstream (it does not bind to blood proteins), and enters the hepatocytes. The affected hepatocytes also exhibit cessation of protein synthesis and cell death, but the symptoms of liver damage (i.e. elevated transaminases) are delayed for up to 48 hours. Within the liver, free amatoxin is bound to bile salts, and is shunted to the gall bladder. Amatoxin contaminated bile is released after a meal, from the gall bladder to the duodenum and back to the liver, causing secondary poisoning. Due to this fact, doctors/veterinarians now recommend that nothing be given by mouth during the course of these poisonings.
In dog models, 80–90% of ingested amatoxins are rapidly excreted by the kidneys; the remaining 10–20% are recirculated through the enterohepatic portal system and back to the liver. In humans, around 40% of amatoxin is rapidly excreted, with approximately 60% remaining in the enterohepatic portal system.
Symptoms of Amatoxin Poisoning
Delayed onset of symptoms, typically followed by a tri-phasic poisoning:
GI Phase (8-24 hours)
Honeymoon Phase (48-72 hours)
Hepatic/Cytotoxic Phase (72-96 hours)
Symptoms are always delayed, although ingestions of multiple mushroom species can complicate the timing of onset. Initial gastrointestinal (GI) symptoms, with severe abdominal pain, vomiting and cholera-like diarrhea, occur anywhere from 6-24 (rarely 36) hours post ingestion. Average time of appearance of symptoms is 8-12 hours post ingestion.
The sooner the appearance of amatoxin poisoning symptoms, the more severe the poisoning.
Amatoxin is primarily eliminated via the kidneys. Dehydration from severe fluid loss during the GI phase seriously impairs the ability of the kidneys to continue to excrete amatoxins, and makes them more susceptible to the direct affects of the amatoxins. Renal failure can occur in the most serious poisonings, and results in a poor prognosis for recovery.
Proper fluid management is essential in an amatoxin poisoning.
Initial GI symptoms, which can mimic the flu in humans, seem to spontaneously resolve, followed by the so-called honeymoon period (up to 24 hours in length), where the patient feels better and is sometimes even discharged from hospital. But don’t be fooled: affected hepatocytes are dying, and any amatoxins that have not been excreted by the kidneys will continue to destroy liver tissue, as they are re-circulated back into the liver via the bile and the enterohepatic circulatory system.
A rise in transaminases usually indicates liver damage, which can progress to complete necrosis, at which point only a liver transplant can save the patient. If the liver damage is only partial, and kidney function can be maintained, recovery and liver regeneration is possible. Although amatoxin can also affect the kidneys, brain, pancreas and testes in severe poisonings, normally this does not occur prior to liver necrosis.
Some mild amatoxin poisonings never progress beyond GI effects. In the most severe poisonings, and over the course of several days, GI bleeding may occur, followed by hepatic encephalopathy, hepatic coma, kidney failure and death.
Heinz Faulstich and Thomas R. Zilker, in the 1994 “Handbook of Mushroom Poisoning, Diagnosis and Treatment,” provide a useful way to judge the severity of amatoxin poisonings, along with likely clinical outcomes. This classification is based on observable symptoms, and not upon concentrations of amatoxin in either blood or urine.
Grade 1: Typical latency period with GI symptoms but no ensuing liver or kidney damage.
Grade 2: Patients with all signs of amatoxin poisoning, with a mild to moderate rise in transaminases (>500 U/L). No clotting dysfunction.
Grade 3: Patients develop severe hepatic damage with a great increase in transaminases, plus impairment of clotting function.
Grade 4: Steep rise in transaminases, steep decline in clotting factors, steep rise in bilirubin and onset of kidney dysfunction.
Grade 1 and 2 need symptomatic treatment only. Grade 3 merits transfer to a liver transplant center. Grade 4 have a poor prognosis in spite of therapeutic interventions.
Bear in mind that “a massive rise in transaminases does not always indicate a fatal outcome; a mild increase in transaminase always indicates a good chance for survival.”
Treating an Amatoxin Poisoning
Amatoxin poisonings are fortunately rare, although it is a fear of this toxin that drives much of the world’s “fungiphobia,” and amatoxin is the toxin to eliminate in any unknown mushroom poisoning case. Ingestions of these can truly be life-threatening emergencies. Most physicians or veterinarians will never see a case.
Many different treatments for amatoxin poisonings have been proposed and used over the centuries; many to most have been discredited. A recent multivariate analysis of thousands of past amatoxin poisonings have shed some light on which chemotherapies work and which don’t (see below). The sooner treatment is started, the better the outcome. Unfortunately, in some cases help isn’t sought until symptoms have progressed. Once multiple organ systems become affected, prognosis is guarded.
Age of the patient, body weight, health, and amounts eaten can all affect outcomes.
According to Faulstich and Zilker, the main aim of any treatment must be to stop the uptake of amatoxins into the liver, either by clearing the portal blood (biliary drainage) or using drugs (chemotherapy) to prevent the uptake of amatoxins.
Biliary drainage (in cases of liver damage) has been used with some success in dogs, and even in some few cases with people. It is an invasive procedure, however, not without its own risks.
During the course of an amatoxin poisoning, bleeding must be prevented; vitamin K and FFP (fresh frozen plasma) are used for coagulopathy. Hepatic encephalopathy (brain dysfunction due to liver insufficiency) can be treated with antibiotics and lactulose, but it usually indicates the urgent need for a liver transplant.
Dr. Michael Beug summarized the current thinking on the best treatments for amatoxin poisoning in his 2015-2016 amatoxin article for NAMA. I quote him below:
"The most important single therapy for amatoxin poisoning is sustained aggressive IV hydration sufficient to maintain strong urine output (amatoxin is primarily excreted in the urine). This fluid therapy involves careful correction of water, glucose, electrolyte imbalances, and acid-base status.
Correction of altered coagulation factors may also be needed (e.g. administration of vitamin K in patients with INR > 2.1).
The retrospective multidimensional multivariate statistical analysis of 2,110 amatoxin poisoning clinical cases published in 2001 indicated that the drugs silibinin, N-acetylcysteine, and putatively ceftazidime {used only in conjunction with silibinin, in only a dozen cases} may be associated with higher rates of patient survival. The multivariate analysis revealed little or no efficacy for Penicillin G, the most frequently utilized drug, and no benefit was found for thioctic acid or steroids. The fact that Legalon-SIL® is an injectable form of silibinin is important because oral absorption of silibinin is poor, making oral ingestion of milk thistle extract of limited utility."
Legalon® SIL is not an “antidote” to amatoxin poisoning, but a way to treat or possibly prevent liver damage. Silibinin/silybin in both oral and intravenous forms have well known hepato-protective effects: silibinin competes with amatoxin for cell entry, and locks the amatoxin binding site; prevents release of amatoxin from affected hepatocytes, reducing the amounts present in enterohepatic circulation; has anti-inflammatory properties; is antifibrotic, allowing the liver to retain its structure, avoiding organ remodeling and hepatic insufficiency; and promotes liver regeneration.
Of the two most likely chemotherapies, Legalon® SIL has shown the most promise, but its current availability is in question. When silibinin is complexed with lecithin in an oral form, it is readily absorbed and its bioactivity is increased. This product is available as a food supplement under the name of Siliphos®. It has not been tested for efficacy in either human or animal amatoxin poisonings, and may be counter-indicated during a strict NPO (nothing by mouth) regime.
History and Current Status of Legalon®
Legalon® SIL was used for decades in Europe, and seemed to have some promise as an adjunct treatment for amatoxin-caused liver damage. In 2006, it was brought to America as an emergency drug by the efforts of a California physician, Dr. Todd Mitchell. In 2009, with Dr. Mitchell as Principal Investigator, and in conjunction with the FDA (North American Food and Drug Administration), FDA clinical trials with Legalon® SIL were begun. The German herbal pharmaceutical company Madaus was sponsor of these trials. Qualifying patients (amatoxin poisoning with documented liver involvement) were treated with Legalon® SIL, in hopes of reducing morbidity (liver transplant) and mortality. Patient enrollment occurred from 2009–2020. Due to only a few cases of amatoxin poisoning being seen each year, it took a long time to build up enough cases for results to be statistically significant. The initial goal was 50 cases; by 2020, 148 cases were treated.
In 2017, Dr. Mitchell exited as Principal Investigator of the clinical trials, and was replaced by Dr. Wallis Marsh. Unfortunately, due to “unobtainable data,” the latest sponsor of this study, Viatris/Mylan Specialty LP, terminated the study in 2020 with no conclusions. Legalon SIL is still available for qualifying human patients via Emergency IND and by contacting Viatris/Mylan.
The current availability of Legalon® SIL in Europe is also under question. Despite its promise, and three decades of standard use in the treatment of amatoxin poisonings in Europe, it is not currently listed in the formulary of the pharmaceutical company Rottapharm (Italy). Rottapharm is the current manufacturer of Legalon® SIL, after a series of international buy-outs starting with Mylan (America) buying out Madaus (Germany) in 2010.
Dr. Sylvie Rapior, one of the authors on that multivariate study on best chemotherapies for amatoxin poisonings, assisted me in attempting to find out if Legalon® SIL was still available in France. A little sleuthing found that currently, despite not being listed in any formulary, Legalon® SIL is available for use in France, but “only with a special authorization (Autorisation Temporaire d'Utilisation/ATU = Temporary Authorization for Use), and is not obtainable without indication validated according to the ANSM (Agence nationale de sécurité du médicament et des produits de santé = National Agency for the Safety of Medicines and Health Products) protocol and without a named patient.” This mirrors the conditions for its use in the US during the clinical trials, where only specific qualifying patients, with existing liver damage from an amatoxin poisoning, were accepted into the trials, and only then was Legalon® SIL sent to the treating physician. In other words, it is possible that no hospital in Europe now has this product on its shelves, ready for immediate use.
I was unable to find confirming data on its current availability in other countries. It does not appear to be available on many of the European pharmacological sites that I searched for online. Neither Rottapharm nor Viatris responded to my inquiries.
AMATOXtest: A Simple, Rapid and Effective Way to Detect the Presence of Amatoxin
Another recent advance in our diagnosis of amatoxin poisonings is the amatoxin point of care (POC) test, or AMATOXtest, created by Candace Bever at the USDA. It can be used on patient urine or on sampled mushroom tissue. It is highly sensitive (and mushrooms make a lot of amatoxins), so care must be taken not to cross-contaminate samples (especially in the case of mushroom sampling). Grossly resembling a home pregnancy or COVID test, a few drops of urine or an extracted mushroom sample causes the test to reveal one or two lines. However, OPPOSITE of a Pregnancy or COVID test, two lines means a negative result, while one line means it’s a positive result. In other words, two lines appear indicating no amatoxins are present (negative result) and only one line (the control line) appears indicating amatoxins are present (positive result). Although it is not currently approved for hospital use, it has been utilized by the veterinary community, and allows rapid diagnoses of amatoxin poisonings, long before liver values start to rise. The earlier that one can confirm an amatoxin poisoning and begin treatment, the better the outcome.
The POC test is very specific. It is very uncommon to experience a false positive (with one exception - phallotoxins are detected by this test). No components in urine nor other chemicals from mushrooms interfere with the test’s ability to detect its target, amatoxins. As many species of mushrooms that produce amatoxins also produce phallotoxins, a false positive from phallotoxins should still be considered very cautiously, despite the differences in their mode of toxicity.
The clinical sensitivity of the POC test is still being investigated. Given the progression of the toxic exposure, the concentration of amatoxins in an intoxicated subject’s urine tends to deplete over time (typically within a few days, but is highly dependent on the total amount of mushroom—and therefore toxin—ingested). As such, a urine specimen taken early (<24hr) after exposure/ingestion will have more reliable results than a sample collected days (>24hr) after ingestion.
.png)
Depictions of the test strips used in this study. (a) Schematic diagram of the lateral flow strip along with a diagram of the reagents on the control line (CL) and test line (TL). (b) A view of the strips when used in a cassette. The left cassette is an example of a sample without amatoxins (negative) and the right cassette is an example of a sample with amatoxins (positive). (i) sample pad, (ii) conjugate pad, (iii) nitrocellulose membrane, (iv) wicking pad, and the arrow indicates the flow direction.
Rapid, Sensitive, and Accurate Point-of-Care Detection of Lethal Amatoxins in Urine, Candace Bever, Creative Commons. doi: 10.1371/journal.pone.0231781
Unfortunately, initial supplies of the POC test were limited. The company sold out, and it is not currently available for sale (8/22). The POC test is now back in production, and projections are for resumed sales some time in October 2022. Interested parties should contact the company directly at that time: https://amatoxtest.com.
Acknowledgements
Thanks to Sylvie Rapior, who helped me track down the current status of Legalon® SIL in France. Thanks to Candace Bever, who made sure that I got everything right about her AMATOXtest. Thanks to Michael Beug, Toxicology Chair for NAMA, who wrote up his own fine amatoxin pages for NAMA in 2016, and has encouraged my work in toxicology for many years. Special thanks to Denis Benjamin, physician, myco-toxicologist and friend, who “checked my work” prior to publication. I appreciate you all.
References
- Angelini, Claudio, et al. 2020. First Report of a Neotropical Agaric (Lepiota spiculata, Agaricales, Basidiomycota) Containing Lethal α-Amanitin at Toxicologically Relevant Levels Frontiers in Microbiology 11:18-33 DOI: 10.3389/fmicb.2020.01833
- Benjamin, Denis R. 1995. Mushrooms, Poisons and Panaceas – A Handbook for Naturalists, Mycologists and Physicians. W.H. Freeman, New York.
- Beug, Michael W. “Amatoxin Mushroom Poisoning in North America 2015-2016,” North American Mycological Association, Toxicology Reports. https://namyco.org/docs/Amatoxin_Poisoning_2015-16.pdf
- Bever CS, Adams CA, Hnasko RM, Cheng LW, Stanker LH. Lateral flow immunoassay (LFIA) for the detection of lethal amatoxins from mushrooms. PLoS One. 2020 Apr 17;15(4):e0231781. doi: 10.1371/journal.pone.0231781. PMID: 32302363; PMCID: PMC7164595.
- Bever, Candace. Personal communication. August 10, 2022.
- Brueckner F, Cramer P. Structural basis of transcription inhibition by alpha-amanitin and implications for RNA polymerase II translocation. Nat Struct Mol Biol. 2008 Aug;15(8):811-8. doi: 10.1038/nsmb.1458. Epub 2008 Jun 13. PMID: 18552824.
- Bushnell DA, Cramer P, Kornberg RD. Structural basis of transcription: alpha-amanitin-RNA polymerase II cocrystal at 2.8 A resolution. Proc Natl Acad Sci U S A. 2002 Feb 5;99(3):1218-22. doi: 10.1073/pnas.251664698. Epub 2002 Jan 22. PMID: 11805306; PMCID: PMC122170.
- Enjalbert F, Rapior S, Nouguier-Soulé J, Guillon S, Amouroux N, Cabot C. Treatment of amatoxin poisoning: 20-year retrospective analysis. J Toxicol Clin Toxicol. 2002;40(6):715-57. doi: 10.1081/clt-120014646. PMID: 12475187.
- Gillessen, Anton, Shmidt, H. 2020. Silymarin as Supportive Treatment in Liver Diseases: A Narrative Review Advances in Therapy 37(1) DOI: 10.1007/s12325-020-01251-y
- Goupil RC, Davis M, Kaufman A, Roberts D, Mitchell T. Clinical recovery of 5 dogs from amatoxin mushroom poisoning using an adapted Santa Cruz protocol for people. J Vet Emerg Crit Care (San Antonio). 2021 May;31(3):414-427. doi: 10.1111/vec.13040. Epub 2021 Jan 17. PMID: 33458945.
- Lüli et al. 2019. Genome of lethal Lepiota venenata and insights into the evolution of toxin-biosynthetic genes.BMC Genomics 20(1) DOI: 10.1186/s12864-019-5575-7
- Poucheret, P., F. Fons, J.C. Doré, D. Michelot, S. Rapior. 2010. Amatoxin poisoning treatment decision- making: pharmaco-therapeutic clinical strategy assessment using multidimensional multivariate statistical analysis. Toxicon, 55(7), pp. 1338-45.
- Spoerke, David and Rumack, B.H. 1994. Handbook of Mushroom Poisoning Diagnosis and Treatment, CRC Press, Inc.
- Sarawi S, Shi YN, Lotz-Winter H, Reschke K, Bode HB, Piepenbring M. Occurrence and chemotaxonomical analysis of amatoxins in Lepiota spp. (Agaricales). Phytochemistry. 2022 Mar;195:113069. doi: 10.1016/j.phytochem.2021.113069. Epub 2021 Dec 26. PMID: 34965486.
© Debbie Viess, August 14, 2022